Solubility is the maximum amount of a solute that can dissolve in a solvent before the solute ceases to dissolve and remains in a separate phase. Water has a very low solubility (is insoluble) in oil, and oil has a low solubility in water. A mixture of water (a polar liquid) and oil (a non-polar liquid) will spontaneously separate into two phases. Phases may also be differentiated based on solubility as in polar (hydrophilic) or non-polar (hydrophobic). As shown in the diagram for iron alloys, several phases exist for both the solid and liquid states. Useful mesophases between solid and liquid form other states of matter.ĭistinct phases may also exist within a given state of matter. Iron-carbon phase diagram, showing the conditions necessary to form different phasesĭistinct phases may be described as different states of matter such as gas, liquid, solid, plasma or Bose–Einstein condensate. However, the state of matter and phase diagram usages are not commensurate with the formal definition given above and the intended meaning must be determined in part from the context in which the term is used. 
Because phase boundaries relate to changes in the organization of matter, such as a change from liquid to solid or a more subtle change from one crystal structure to another, this latter usage is similar to the use of "phase" as a synonym for state of matter. Also, the term phase is sometimes used to refer to a set of equilibrium states demarcated in terms of state variables such as pressure and temperature by a phase boundary on a phase diagram. The term phase is sometimes used as a synonym for state of matter, but there can be several immiscible phases of the same state of matter. The glass of the jar is another separate phase. In a system consisting of ice and water in a glass jar, the ice cubes are one phase, the water is a second phase, and the humid air is a third phase over the ice and water. A simple description is that a phase is a region of material that is chemically uniform, physically distinct, and (often) mechanically separable. 
: 86 : 3 Examples of physical properties include density, index of refraction, magnetization and chemical composition. 
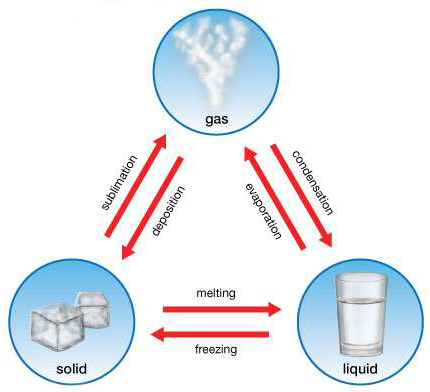
Less energy is needed to allow molecules to move past each other than to separate them totally.In the physical sciences, a phase is a region of space (a thermodynamic system), throughout which all physical properties of a material are essentially uniform. The enthalpy of vaporization of a given substance is much greater than its enthalpy of fusion because it takes more energy to completely separate molecules (conversion from a liquid to a gas) than to enable them only to move past one another freely (conversion from a solid to a liquid). Substances with high boiling points are those with strong intermolecular interactions that must be overcome to convert a liquid to a gas, resulting in high enthalpies of vaporization. The substances with the highest melting points usually have the highest enthalpies of fusion they tend to be ionic compounds that are held together by very strong electrostatic interactions. \): Melting and Boiling Points and Enthalpies of Fusion and Vaporization for Selected Substances.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
